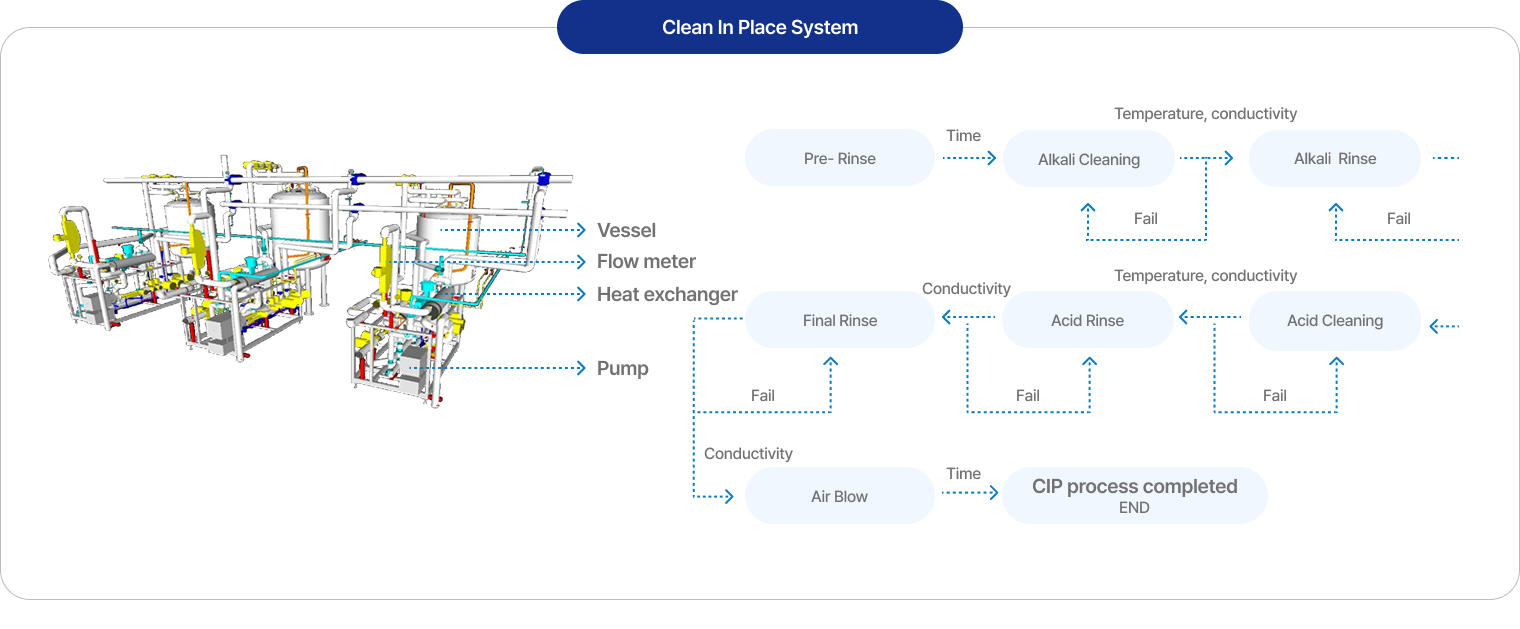
Bio/pharmaceutical field 




CIP (Clean In Place)
In 2014, we participated in the localization project for overseas imported cleaning technology at Samsung Biologics Plant 2, securing equipment manufacturing and validation technology. Following the completion of localization, we have delivered over 200 units to major pharmaceutical companies including Samsung Biologics, Polus, and Hanmi Pharmaceutical.Core features